[Front Page] [Features] [Departments] [SGAP Home Page] [Subscribe]

Short Cuts
Readers are invited to submit short items of interest about Australian plants to be included here. If submitting non-original material (eg newspaper or magazine cuttings), please also advise if the author has given permission to republish and, if not, please provide a contact address so that permission can be sought.
Short Cuts in this issue:
 From Seed to Tree From Seed to Tree
- A better method for establishing successful trees
 Tea Tree Oil - First Aid in a Bottle Tea Tree Oil - First Aid in a Bottle
- No, it's not snake oil! This stuff really works!
 The "Bog" Method.... The "Bog" Method....
- Funny name....but a great way to germinate seed
 Viruses and Orchids Viruses and Orchids
- Care needed to avoid spreading of infection
 Eremophila racemosa - Back from the Brink! Eremophila racemosa - Back from the Brink!
- A new population of this rare Emu Bush
 Eight Ways to Kill Cuttings! Eight Ways to Kill Cuttings!
- Stupidity is never far away in this list of things not to do

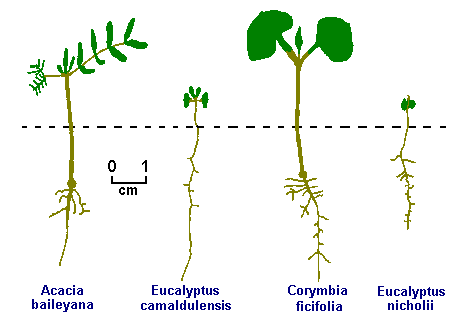
From Seed to Tree
Fon Ryan takes a new look at the root system on developing seedlings and decides that there is a better way....
Over the past twelve years I have spent much of my spare time in researching the propagation from seed, planting out and growth of Australian native shrubs and trees. This research has included digging up scores of 'failed' tress in order to clean and examine the roots.
The first transplanting
In conventional seed propagation, germination takes place in shallow containers, then the seedlings are transplanted from these containers into individual tubes or pots. This transplanting operation is the cause of subsequent failures in a percentage of trees after they are planted out into the ground. If it is not done with care, faulty roots will usually result and this leads to future unthriftiness, slow growth rates, looseness in the ground for extended periods, toppling or death. These symptoms may take from one to ten years to appear. Even if transplantation from seed container to pot is done with care, its inherent difficulty may still create a faulty root structure.
 |
| |
|
Diagram showing the natural root development of seedlings 14 days after emergence of the cotyledons. Note the development of the laterals. This quickly established root structure will be the basis for the tree's future stability. |
Poor transplantation frequently results in the tap root getting a small loop in it with the usual result that as the root thickens the loop tightens like a knot. This strangles the root by cutting sap flow around most of its circumference. Strangulation will also follow if all, or most, of the roots get interwoven at this initial seedling transplantation. Pot bound roots also lead to failures similar to those caused by poor seedling transplantation - and sometimes in conjunction with it.
Occasionally, one lateral root (usually just above the mass of tangled roots) escapes being strangled or obstructed. It may then 'take over' and adequately supply the tree with water and nutrients. In one such case a Eucalyptus nicholii grew to a height of 5m in ten years, then gradually fell over, pivoting around the horizontal axis of that one root. It is important to understand first, that seedling transplantation root faults are usually not evident at the planting out stage of the seedling; and that second, that noting can be done to rectify such faults.
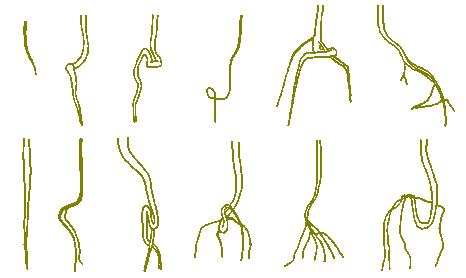 |
|
Diagram showing the roots on a random selection of commercially grown15cm tube stock. |
A better way
To overcome all these problems I recommend the following method:
- Use a pot with a minimum depth of 150mm and preferably a minimum diameter of 80mm.
- Fill the pot with the growing medium to within about 10mm of the top. Use enough seeds to obtain one healthy seedling. Sow the seeds around the centre of the pot. Shortly after germination, remove and discard any excess seedlings. There must be no transplanting into or out of this pot and no repotting as the seedling grows.
- Before any roots have reached the bottom of the pot, stand it on raised open mesh or a rack. This helps prevent the roots from growing out through drainage holes.
- When planting the seedling into the ground: remove it from the pot and then cleanly cut the bottom 20mm of soil and tangled roots from the root/soil ball (This includes cutting the tap root). Also shave 5mm of soil and roots from the outer surface (side) of the roots/soil ball. The roots will grow on from where they were cut.
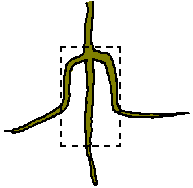 | |
| |
| Dried root specimen from a tree grown by Fon's method and dug up after successful establishment in the ground. The tap root had grown straight on after being cut at planting time. Laterals had resumed horizontal growth after restriction in the pot, demonstrating the need to trim the sides of the root ball as well as the base (pot size 80mm x 200mm). |
From the September 1989 issue of the newsletter of the Victorian Region of SGAP. (based on a talk given to the Central Highlands Group.)
[ Return to Index ]

Tea Tree Oil - First Aid in a Bottle
Eucalypts are not the only Australian native plants to yield a good oil; tea-tree oil has long had a reputation as a useful antiseptic substance. Marianne Stagg describes some of its features.
Tea Tree Oil is derived from one species of Melaleuca, (M.alternifolia). Although the species grows naturally in only one region in the world, between Grafton and Murwillumbah on the north coast of New South Wales, it is not uncommon and exists in large areas. As early as 1770, M.alternifolia attracted attention. When exploring the above area, Captain James Cook wrote how he collected leaves from what he described as "Tea Tree" and boiled them to make a spicy tea. Sir Joseph Banks, who was on the expedition with him, collected samples of the same leaves and took them back to England for further study.
One hundred and fifty years later in 1923, an Australian Chemist, Dr A R Penfold discovered that not only was the pale lemon tinted oil distilled from the tea tree leaves pleasant smelling, it was also non-poisonous, non-irritant and had exceptional antiseptic and fungicidal properties whilst in its purist form. In 1933, journals such as the Australasian Journal of Pharmacy, stated that "the oil is a powerful disinfectant,... and has been used successfully in a wide range of septic conditions."
During World War 2, tea tree oil was so popular it was an essential item for every first aid kit issued to army and naval units in tropical regions. Demand however eventually surpassed supply and synthetic alternatives gained popularity.
By the 1970s concern over possible side effects from toxic substances and synthetic medicines rekindled interest in natural therapies and stimulated further research into the medicinal value of tea tree oil. Subsequently, a world interest in the oil led to the formation of the Australian Tea Tree Industry Association, the establishment of many plantations and the development of a multi-million dollar industry.
For therapeutic purposes, only authentic antiseptic grade Melaleuca alternifolia should be used. Uses for tea tree oil include:
- Use neat or diluted in water to treat mouth ulcers, cold sores, sprains, boils, bites, leeches, ticks, pimples, burns and warts.
- Five drops to a glass of water makes a gargle or mouthwash for sore throats, sore gums and bad breath.
- A few drops added to humidifiers or vaporisers relieves nasal and chest congestion.
- Ten drops in bathwater provides relief of muscle aches.
- As a mixture of 1 part tea tree oil to 10 parts of almond oil, it can be used to relieve arthritic pain and heal cuts, abrasions, dry skin, dermatitis and cradle cap.
- Ten drops of oil added to 250ml of human shampoo improves dry or oily hair and itchy scalp, and treats head lice. Added to pet shampoo it kills fleas, and soothes rashes, itches and cuts.
From the ASGAP Newsletter, October 1996 (originally in the newsletter of SGAP's North West Tasmania Group, September 1996).
[ Return to Index ]

The "Bog" Method
Preventing fine seeds from drying out in seed beds can be a problem. Alf Salkin has the answer.....Enter, "the Bog Method".....
The first rule is that the seed should not be sown deeper than its own minimum dimension, so that fine seed such as Brachyscome, Eucalyptus, Melaleuca or Stylidium is actually sown on the surface and not covered. This presents two problems; both of them associated with water. The first is the problems of keeping the seeds moist. The second is preventing the seed from being washed away by heavy rain or watering. Both of these problems can be overcome by a technique called the Bog Method.
In this method a 50 mm (2") new or sterilised pot is filled with a soilless mix - I use six parts river gravel to one part peat moss filled right to the top. The reason for the 50 mm pot is that more than 100 seedlings will germinate in it. If you want more than this use a bigger pot or preferable a multiple of 50 mm pots. Pots are stood in a container of water which is no deeper than half of the depth of the pot. When the water has thoroughly wet the mixture by capillary action, the seed is sown finely on the surface. With most seed, the very small cotyledons can be seen within two weeks.
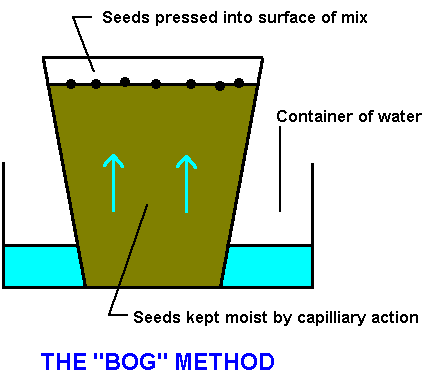
One advantage of this system is that the pot can be kept indoors and be inspected frequently: but once germination has commenced the pot should be placed in the open, otherwise the young plants will elongated and be weak. The technical term for this is "drawn".
The general rule for transplanting is to do this when the plant has produced its second set of leaves but, in practice, with numerous very small seedlings, some will have produced a number of set of leaves while others are still at the cotyledon stage. So whether you wait for all the seedlings to reach the true two-leaf stage will depend on whether you want every seedling or can afford to waste some.
To transplant these tiny seedlings the pot is shaken sideways over a tray so that the tops of the seedlings are at one side and the mixture and the roots at the other. By lifting the stalk and leaves of the uppermost seedling and gently pulling the young plant with its long root, it may be separated from the mixture and then potted up in a previously prepared potting mix. This sort of work should be undertaken on a cool, moist, windless day or in place with these sort of humid conditions, as the young seedlings will have suffered some damage to their microscopic root hairs even if no damage is observable to the naked eye.
One reason why the place where plants are raised is called a nursery is that young plants need special care - nursing. Transplanting for the young seedling is like you having an operation on your stomach. The feeding areas have been disrupted and even with the greatest care some damage is still done. The procedure for this after-care is known as hardening off. In this technique the plant is gradually moved from the glasshouse or cool frame to a shade area and finally out in full sun.
One final thing about transplanting - it is not a good idea to pot seedlings in large pots in the hope that they will grow quicker. They won't. The reason is that without the plant's roots actually growing vigorously, the mixture will become sour (acidic) and hinder growth. I generally use 50 mm pots and open friable mix with up to 50% propagating sand when transplanting seedlings grown by the Bog method. This method of sowing seed can be done on a window sill and some children find it fascinating: but it is not for those who require instant gratification. The process from germination to garden plant can take as long as 12 months.
From the September 1985 issue of the newsletter of the Victorian Region of SGAP.
[ Return to Index ]

Viruses and Orchids
Mitch Harris explains why care is needed when handling orchids. Viral infections can be inadvertently transferred from one plant to another by a careless grower.
Twenty-five viruses have been found to infect orchids around the world. Not all these viruses are in Australia. The two most common are Odontoglossum Ring Spot Virus (ORSV) and Cymbidium Mosaic Virus (CyMV). Virus are very small plant pathogens that can only be detected by an electron microscope, magnified tens of thousands of times. ORSV is known to be viable for up to seven months in dried sap!
Diagnosis
Leaf symptoms are extremely variable depending on what genus of orchid is infected with which virus.
- Red lesions, chlorotic areas, brown necrosis surrounding bleached tissue, and interveinal chlorosis are symptoms of ORSV.
- Twisting of leaves, brown necrotic colour breaking and raised brown necrotic lesions are symptoms of CyMV.
Transmission
Viruses can be transmitted in several ways:
- in sap, i.e. when sap from an infected plant comes in contact with a healthy plant. This is most commonly done by mechanical or physical means (i.e. secateurs or by hand)
- by insect (e.g. aphids, Dendrobium beetles)
- by pollen transfer
- during vegetative propagation of many plants
Control
Follow a few simple procedures so that a virus does not spread throughout your collection.
- Diagnosis - is this unusual symptom caused by a virus or not?
- Sanitation - all cutting tools, benches, pots and your HANDS need to be sterile during work with your orchids.
Summary
- Keep tools and benches clean. Use clean paper on benches for each plant repotting procedure.
- Wash hands between each repot.
- Destroy virus-infected plants.
- Do not re-use potting media.
- Control insects.
- Space plants out on benches.
- Avoid handling plants.
References
- Virus Disease of Orchids; Orchids Australia
- Pest and Disease Handbook; American Orchid Society
- Dendrobium Orchids of Australia; W.T. Upton
- The Orchid Growers Manual; G.C. Morrison
- Dendrobium kingianum; P.B. Adam and S.D. Lawson
- Tropical Orchids of Australia. P.S. Lavarack and B. Gray
From the December 1996 issue of the newsletter of the North Shore District Group of SGAP. Mitch works for Ku-ring-gai Council in Sydney.
[ Return to Index ]

Eremophila racemosa - Back from the Brink!
A discovery of a large population of this rare Emu Bush may give it a better chance of survival. Bob Chinnock has the story and explains why fire is an important factor in the decline and recovery of this and other species.
Quite a few of the eremophilas that are widespread in cultivation like E.nivea, E.microtheca and E.racemosa are extremely rare and endangered in the wild. The latter species, for example, was known only from a small area east of Hyden, Western Australia, where it occurred on disturbed roadside verges up to 15km from a road junction. Although I have visited the area on about seven occasions in the last 20 years I have never located E.racemosa growing in natural undisturbed sites. Furthermore it is rarely found in populations of more than 5 or 6 plants so Guy Richmond and I were delighted in 1993 when we found a "large" population of 38 plants.
As it turned out this population was subsequently destroyed early the next year when a massive fire passed through the area followed soon after by road widening. Regeneration in the burnt areas following the 1994 fire have been spectacular. With the removal of the mallee canopy and tall understorey shrubs, small plants like Eremophila, Anthoceris, Symonanthus and various Myrtaceae, grasses and ephemerals have come up in abundance.

Eremophila racemosa is a species with a very restricted occurrence in nature but which is reasonably well established in cultivation. Select the thumbnail image or plant name for a higher resolution image (26k).
Early in 1996 Guy was out in the area and found a population of E.racemosa which he estimated at between 1 and 2 million. He also brought back a vegetative eremophila which I could not identify.
On my return trip to Australia at the beginning of October I stopped off in Perth and Guy and I did a rapid trip to the area to see the E.racemosa population and to locate the unidentified eremophila. We could not get into the large racemosa population because of a flooded track but we did find another small one of about 5,000 plants. Most of the plants were in late bud and there was quite a lot of colour variation. The yellow/cream variant recently introduced into South Australia was seen occasionally.
When we got to the site of the unidentified eremophila we found it in full flower. It turned out to be a new species which I had seen previously at Kings Park a couple of years earlier. When I was shown it at Kings park I was non committal. I did not know whether it represented a single plant found in the wild (in which case it could be a hybrid) or one plant collected from a population. The population found consisted of many thousands of young plants up to 1m high which had come up following the fire.
It is closely related to E.viscida with large shiny leaves, and yellow unspotted flowers and sepals. Very occasionally in the population we encountered a carmine spotted flower form which is much more striking. A number of plants are now establishing in Adelaide. It is likely that this species which I have tagged E."Lucida" will probably go straight on to the the Rare and Endangered priority 1 list.
Other eremophilas were also abundant throughout the burnt out areas. E.subfloccosa which I have only seen as scattered plants in South Australia and Western Australia was up in the tens of thousands. I counted up to 16 plants per square metre. E.serpens was likewise in the thousands at one location and two subspecies of E.densifolia were forming dense ground covers at various locations.
What can we learn from this fire? Eremophilas like E.racemosa, E. "Lucida", E.densifolia, E.subfloccosa and E.serpens are fire response species which, after fire, regenerate in large numbers. After the canopy species recover and regenerate their canopies, species like these eremophilas will rapidly decline in numbers to small scattered populations and persist in disturbed sites on roadsides, tracks, clearings until the next major event occurs, resulting in their resurrection.
From the December 1996 issue of the newsletter of the Eremophila Study Group. Bob Chinnock is a botanist at the herbarium of the Adelaide Botanic Gardens.
[ Return to Index ]

Eight Ways to Kill Cuttings!
"How shall I dispatch thee?...Let me count the ways!"
There are far more than eight ways to stuff up a batch of cuttings but Ross Doig doesn't want to discourage you too much.....
This short article is by no means an exhaustive account of the subject which, as many a reader will know, is capable of considerable expansion. Enough will be said, I hope, to be of interest and instruction.
First - dry out foliage or medium
In order to achieve this, allow the cover to blow off the cutting box: inspect plastic covered cutting pots and forget to replace cover on one: leave door of glasshouse/shade house open on a day of good drying westerly winds: put cutting box out to get morning sun, preferable on a day when the temperature will reach 25 degrees C or more and, for full effect, open cover a little to ventilate. Remember late in the afternoon to replace in shade.
Second - overwater (or when is enough too much?)
The correct answer under this heading is when grey fungus, mildew, collar rot, stem rot, flourish. Be sure to keep everything soggy in a small cutting box: maintain a low light level in cutting box or glasshouse and combine with excessive mist or spray in winter and mid summer: cover cutting boxes in shade with shadecloth and water daily making sure that external humidity is high.
Third - the hairy and/or herbaceous
Cuttings in this category may be terminated by the methods cited in the second way above. But add - bury tubes or pots in damp (soggy preferred) sand; peatmoss, coco peat. Use plenty of peatmoss in cutting medium: keep hairy leaves and stems damp at all times. Actinotus helanthii and Olearia species respond very well to these treatments.
Fourth - stem clasping leaves
Select cuttings of such material particularly from the Epacridaceae family and make sure that slivers of bark are removed along with the leaves. Death occurs quite rapidly. While not exactly in this category but having similar effect is to use blunt scissors or knife to drag or crush rather than cut cleanly the base of the cutting. The mangled end, while more aesthetically pleasing, ensures that the cutting has no future.
Fifth - leaves how many?
Take literally the advice to leave a few leaves, particularly when leaves are below 2 centimetres in length, leaving no margin for error. Banksias are ideal for this experiment with say three or four leaves as they gradually change colour and die one by one exhibiting autumn tints which are quite outstanding. I'm never game to leave less than 20 with B.ericifolia or B."Giant Candles" and 10 or so with the Waratah. Melaleucas are notable for leaf drop especially with those slow to take root and held over the winter so with this genus leave as many as possible.
Sixth - summer heat
A simple killing field system. Spray or mist several times a day, keep ventilation to a minimum and make sure the cutting box/shade house environment is as soggy as possible. Then treat with fungicide in an attempt to inhibit growth while knocking off fungus and mildew.
Seventh - low light levels
My greatest barrier to successful propagation in early attempts was lack of light. A shed with sunlight from the north-east for two hours in the morning provided the only light. All medium mixtures, small or large boxes with plastic or shade covers, or open boxes on benches, were to no avail. In six months total failures with over 100 sets of cuttings. Success came only when boxes were moved outside into a semi shaded situation.
Eighth - the unkindest cut of all
Unkindest because the damage can't be seen and the cuttings remains (apparently) viable, above ground for up to two years. Upon inspection the last 10 - 12 millimetres of the stem is black- obviously dead. Rarely roots form at the node above. Not all cuttings are affected, some take root as normal. Boronias, grevilleas, zierias, tetrathecas and hakeas demonstrate that a wide range of genera is susceptible. The phenomenon occurs with and without the use of hormones and I have no explanation as to the cause.
Having discouraged you all with the bad news (120 sets of cuttings in six months sent to happy hunting grounds) I come to the good news. Between November 1991 and October 1993 I have struck 395 species of Australian natives from a wide range of genera with very few failures.
From the July 1994 issue of the newsletter of the Parramatta and Hills District Group of SGAP.
[ Return to Index ]
 [Front Page] [Features] [Departments] [SGAP Home Page] [Subscribe]
[Front Page] [Features] [Departments] [SGAP Home Page] [Subscribe]
Australian Plants online - September 1997
The Society for Growing Australian Plants
|